Chemistry Solutions
Teacher instructions and preparation
 Not every student is going to love chemistry. Not every student is going to remember all or even the majority of his or her high school chemistry course. Not every student is going to become a chemist, major in chemistry, or even perhaps take a chemistry course in college. But many, if not most, high school students take chemistry. Therefore, I want to make sure that by the end of my courses, each student in my 10th-grade chemistry class knows the importance of chemistry in everyday life. Chemistry is everywhere. It has had a tremendous impact on history and continues to impact our lives. To open my students’ eyes to this idea, I have two activities that I incorporate each year into my chemistry course. First, students read Napoleon’s Buttons: How 17 Molecules Changed History (1), accompanied by in-class discussion and a writing assignment. I also have students participate in an activity called Molecule of the Week. Students find learning about the molecules fun and interesting, and they leave the class with an appreciation for the relevance of chemistry in their lives.
Not every student is going to love chemistry. Not every student is going to remember all or even the majority of his or her high school chemistry course. Not every student is going to become a chemist, major in chemistry, or even perhaps take a chemistry course in college. But many, if not most, high school students take chemistry. Therefore, I want to make sure that by the end of my courses, each student in my 10th-grade chemistry class knows the importance of chemistry in everyday life. Chemistry is everywhere. It has had a tremendous impact on history and continues to impact our lives. To open my students’ eyes to this idea, I have two activities that I incorporate each year into my chemistry course. First, students read Napoleon’s Buttons: How 17 Molecules Changed History (1), accompanied by in-class discussion and a writing assignment. I also have students participate in an activity called Molecule of the Week. Students find learning about the molecules fun and interesting, and they leave the class with an appreciation for the relevance of chemistry in their lives.
Napoleon’s Buttons: How 17 Molecules Changed History
 The summer before they take chemistry, my students read Napoleon’s Buttons. The introduction of the book explains chemical structures and gives examples of skeletal structures of organic molecules. It doesn’t provide an in-depth picture of covalent bonding, but it gives the reader enough background to understand what is depicted in a molecular structure. The subsequent 17 chapters of the book describe various molecules, or classes of molecules, that have “changed history,” including ascorbic acid , “the pill,” and chlorocarbon compounds. Throughout the first week of class, I facilitate a discussion of the book. For our schedule, this is two to three 50-minute class periods. I start with the simple question, “What is a molecule or class of molecules that you found interesting, and why?” Then I move into, “What did you learn from this book? What do you agree or disagree with?” Depending on the size and population of the class, the discussions vary in depth and length, but the students tend to be participatory. As long as they have read a few chapters of the book, students usually have some insight to share. I find the students are more excited to discuss Napoleon’s Buttons than other summer reading books I have assigned. They also bring up molecules or ideas from the book later in the semester, which I have not encountered with other summer reading.
The summer before they take chemistry, my students read Napoleon’s Buttons. The introduction of the book explains chemical structures and gives examples of skeletal structures of organic molecules. It doesn’t provide an in-depth picture of covalent bonding, but it gives the reader enough background to understand what is depicted in a molecular structure. The subsequent 17 chapters of the book describe various molecules, or classes of molecules, that have “changed history,” including ascorbic acid , “the pill,” and chlorocarbon compounds. Throughout the first week of class, I facilitate a discussion of the book. For our schedule, this is two to three 50-minute class periods. I start with the simple question, “What is a molecule or class of molecules that you found interesting, and why?” Then I move into, “What did you learn from this book? What do you agree or disagree with?” Depending on the size and population of the class, the discussions vary in depth and length, but the students tend to be participatory. As long as they have read a few chapters of the book, students usually have some insight to share. I find the students are more excited to discuss Napoleon’s Buttons than other summer reading books I have assigned. They also bring up molecules or ideas from the book later in the semester, which I have not encountered with other summer reading.
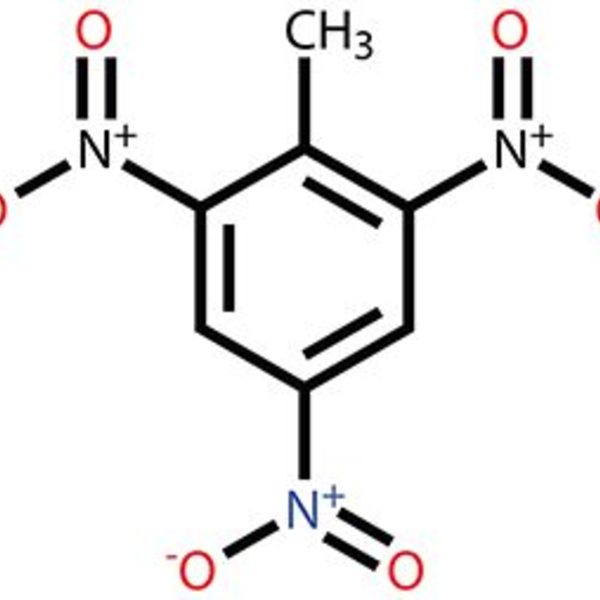 As the class discussion starts to dwindle, I draw structures of molecules from the book on the board. I review skeleton structures and explain why not every carbon and hydrogen atom needs to be explicitly drawn and labeled. I do not discuss every molecule from the book but instead choose a few of the classes and types that I think students might find most interesting. I start with benzene, showing the aromatic rings and conjugated systems. This leads to derivatives such as phenol, toluene, TNT , and aspirin. I also introduce sugars, showing the difference between glucose, fructose, sucrose, and cellulose. I tell students upfront that I do not expect them to memorize all of these molecules. Instead, I want them to know that real-life materials are made up of molecules and I want them to recognize classes of molecules such as aromatics and sugars.
As the class discussion starts to dwindle, I draw structures of molecules from the book on the board. I review skeleton structures and explain why not every carbon and hydrogen atom needs to be explicitly drawn and labeled. I do not discuss every molecule from the book but instead choose a few of the classes and types that I think students might find most interesting. I start with benzene, showing the aromatic rings and conjugated systems. This leads to derivatives such as phenol, toluene, TNT , and aspirin. I also introduce sugars, showing the difference between glucose, fructose, sucrose, and cellulose. I tell students upfront that I do not expect them to memorize all of these molecules. Instead, I want them to know that real-life materials are made up of molecules and I want them to recognize classes of molecules such as aromatics and sugars.
The next progression of the lesson is a writing assignment. They choose three molecules from the book, and for each molecule, they write one or two paragraphs summarizing the authors’ point of view of the importance of the molecule. Then they write one to two paragraphs about whether they agree or disagree with the authors’ view. I ask them to come up with two molecules that were not included in the book and write one to two paragraphs about their importance, including why each can be considered a “molecule that changed history.” I do not provide them with suggestions of sources for their molecules, but I will help them if they ask me. No one has ever approached me for help finding a molecule; students tend to be resourceful and creative in finding molecules on their own.
Keep in mind this is the first week of class for 10th graders in their first chemistry class, so answers vary tremendously. Each year, I have several students argue the importance of water. Others go with explosives or biological molecules or drugs (both of the legal and illegal variety). But there are always several students who submit really unique molecules, such as captopril (a drug for hypertension) and Kevlar (the material in bullet-proof vests). This activity provides an interesting insight into the chemistry background and the interest of the students. The complete lesson and grading rubric can be found in the AACT resource library.
Molecule of the week
Napoleon’s Buttons introduces students to chemistry and allows me to teach the basics of drawing molecules. I want to keep building on those concepts, even though the first chapter in the textbook is about significant figures and unit conversions. I want students to continue seeing and learning about molecules, no matter what chapter they are in. Therefore, I take about 10 minutes each week to introduce the Molecule of the Week. This isn’t a novel concept; there are websites dedicated to this idea and many others teachers and professors use similar activities. Students enjoy learning chemistry in this way and begin to realize through this exercise that chemistry really is everywhere.
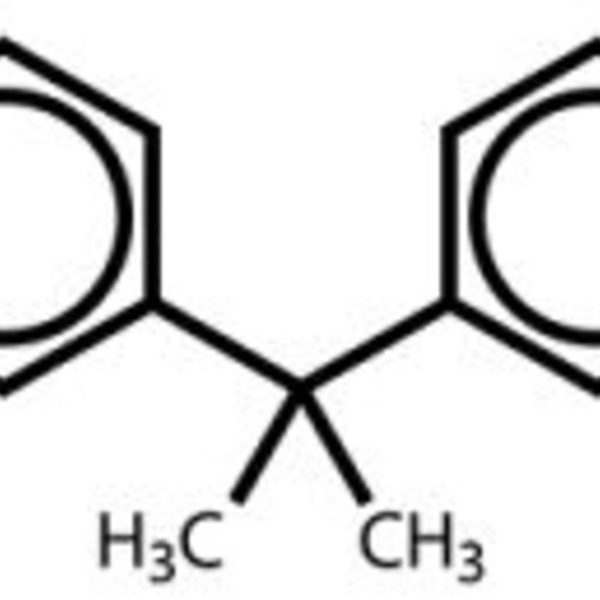 The molecules I choose change each year, but some of my favorites are bisphenol A (BPA), monosodium glutamate (MSG), and butylated hydroxytoluene (BHT). These are all acronyms many students have heard, but they don’t necessarily know what the molecule looks like or why they hear so much about the chemicals. Around Halloween, when glow sticks appear on the shelves of local convenience stores, I introduce them to diphenyl oxalate and the reaction that makes a glow stick glow. In the fall when the leaves change, I introduce conjugated molecules such as chlorophyll and lutein. Sometimes I choose molecules that have recently appeared in the news or that students ask about in class.
The molecules I choose change each year, but some of my favorites are bisphenol A (BPA), monosodium glutamate (MSG), and butylated hydroxytoluene (BHT). These are all acronyms many students have heard, but they don’t necessarily know what the molecule looks like or why they hear so much about the chemicals. Around Halloween, when glow sticks appear on the shelves of local convenience stores, I introduce them to diphenyl oxalate and the reaction that makes a glow stick glow. In the fall when the leaves change, I introduce conjugated molecules such as chlorophyll and lutein. Sometimes I choose molecules that have recently appeared in the news or that students ask about in class.
For each molecule, I share its structure and any notable functional groups. I discuss what makes it interesting, whether it is its history, chemistry, or current relevance. I encourage students to pay close attention and take notes, and I use the information about the molecules as bonus questions on tests or quizzes. I try not to overload them with information about each molecule, but instead I want them to realize that chemistry is interesting as well as pertinent to their lives.
In the fall semester, I choose a different molecule each week. But at the beginning of the spring semester, as a homework assignment, students research and present a molecule of their choice. They are assigned a date to present, one student per week. The day before their presentation, they submit their molecule’s name, molecular formula, molecular structure, and information they are going to share, including at least two references. During class, they have 10 minutes to share their molecule’s name, molecular structure, and facts about its relevance to life. The full lesson is available in the AACT resource library .
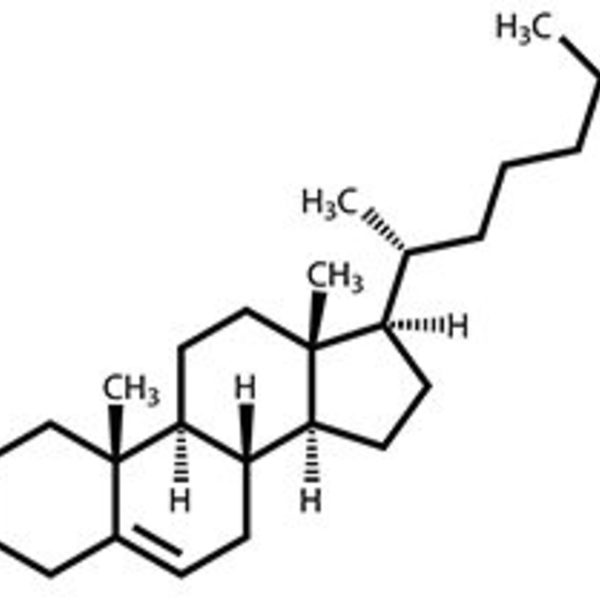 Students’ choices have ranged from DEET to cholesterol , from dopamine to AZT. I was thoroughly impressed when one of my weaker students did a stunning presentation on tetradotoxin (the neurotoxin in pufferfish) and gave a detailed explanation of sodium ion channels. Another student presented the pesticide DDT and included a passionate overview of its harmful effects to the environment that would have made naturalist, advocate, and writer Rachel Carson proud.
Students’ choices have ranged from DEET to cholesterol , from dopamine to AZT. I was thoroughly impressed when one of my weaker students did a stunning presentation on tetradotoxin (the neurotoxin in pufferfish) and gave a detailed explanation of sodium ion channels. Another student presented the pesticide DDT and included a passionate overview of its harmful effects to the environment that would have made naturalist, advocate, and writer Rachel Carson proud.
A few students supplement their talk with short videos from the Internet, but most do “chalk talks,” where their classmates copy the structure and notes from the board. The presenter allows students to ask questions about the molecule, and this often leads to engaging conversations among students. Through these presentations, I have been introduced to many interesting molecules that I use in subsequent years. It is wonderful to see the creativity and excitement students have for this assignment, both during and after.
It’s not uncommon for upperclassmen to stop by to see what this week’s molecule is. If it is one they haven’t learned about, they usually demand to know its importance. And I’ve had students share stories with me about a molecule they talked about with their family at the dinner table or one they heard about in the news or on the Internet. I love that the students are still excited about relevant and interesting molecules. I hope they will hold onto that excitement for chemistry wherever they go in their lives.
Do not reinvent the wheel
There are many websites that provide information about molecules and their interesting chemistry. Some of my favorites are the American Chemical Society’s "Molecule of the Week Archive," Andy Brunning’s blog "Compound Interest," and Bristol University’s "The Molecule of the Month" site. The Chemical Abstracts Service website is also helpful, as they provide general information about a molecule and an abstract from a scientific journal article pertaining to the molecule. There are also books with collections of interesting stories, and two of my favorites are The Disappearing Spoon (2) and The Poisoner’s Handbook (3). I have included a list of some of the molecules that my students or I have presented in the past in the lesson about the molecule of the week.
I encourage chemistry teachers to incorporate relevant molecules into their chemistry course. Napoleon’s Buttons is an excellent read for both students and teachers, and the molecule of the week is a way to introduce students to a variety of interesting molecules. The associated assignments provide opportunities to incorporate writing, researching, and presenting in the chemistry curriculum. These activities take minimal classroom time but are invaluable because they have a lasting impact on students’ education and engagement in the course.
- Le Couteur, P. and Burreson, J. Napoleon’s Buttons: How 17 Molecules Changed History; Jeremy P. Tarcher/Penguin: New York, 2004.
- Kean, S. The Disappearing Spoon and Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the Elements; Little, Brown and Company: New York, 2010.
- Blum, D. The Poisoner’s Handbook: Murder and the Birth of Forensic Medicine in Jazz Age New York; The Penguin Press: New York, 2010.
Photo Credit: Penguin Putnam (Top), Depositphotos.com/electropower (Remaining)